How to Choose High-Quality Epimedium Extract Powder?
2026-02-28 13:35:47
It's important to carefully consider the standardized icariin content, third-party testing certifications, and supplier reliability when picking the right epimedium extract powder to make sure it meets regulatory requirements and works well in nutraceutical, pharmaceutical, and functional food formulations.
This complete guide talks about the rising need for epimedium extract powder around the world among industrial buyers, original equipment manufacturers (OEMs), and wholesalers. It is very important to choose high-quality powder to make sure that the product works, that it follows the rules, and that customers trust the nutraceuticals and health-related businesses. The market for botanical extracts has grown a lot, and epimedium derivatives are getting a lot of attention because they can be used in a lot of different ways, such as in male vitality supplements, bone health formulations, and functional drinks.
Understanding Epimedium Extract Powder and Its Market Value
A key ingredient in current nutraceutical formulas is epimedium extract powder, which is mostly made from Epimedium brevicornum Maxim and related species. People love this plant extract because it contains the bioactive substance icariin, which is used to standardize and rate the quality of products. The extract is very useful in many different types of industries because it can be used in both male and female health products and has clear physiological benefits that are backed up by clinical study.
The world market for epimedium derivatives shows strong growth patterns, with the strongest ones being in Europe, North America, and the Asia-Pacific region. The market is being driven by growing health trends, more consumers knowing how effective natural ingredients are, and more people wanting standardized botanical alternatives to synthetic compounds. Analysts in the industry say that the herbal extract segment has been growing steadily year over year. Epimedium continues to hold a leading place thanks to its proven safety profile and bioactivity.
Primary Applications Across Industries
There is a huge range of manufacturing situations where epimedium extract can be used. In solid dose nutraceuticals, the powder is one of the main active ingredients in products that boost libido and testosterone. Manufacturers often mix the extract with ingredients that work well together, such as L-Arginine or Tribulus terrestris, to make complicated formulations that work better together.
For functional beverage uses, specific water-soluble grade versions or nano-emulsification technologies are needed to keep the mixture from settling and hide the bitter taste that comes with having a lot of flavonoids. When making energy drinks and pre-workout mixes, where taste and clarity are still very important, these technical issues become even more important.
Utilizing icariin's known ability to increase osteoblast proliferation, bone health products are a new area of use. Because these uses last a long time and bone health claims are closely watched by regulators, they need strict heavy metal control procedures.
Market Positioning and Competitive Advantages
To understand market positioning, you need to look at epimedium's competitive benefits compared to other botanical ingredients in great detail. The standard icariin content of the extract gives formulators accurate bioactivity levels that let them make accurate dosing calculations and ensure uniform performance from batch to batch. This ability to standardize sets quality sellers apart from commodity providers, whose potency levels may vary.
Because the ingredient is appealing to both men and women, it opens up new market possibilities beyond traditional male health categories. This lets companies make crossover products that appeal to a wider range of people. Research showing benefits for bone density and cardiovascular health opens up more formulating options for creative product developers looking for unique ways to place their products.
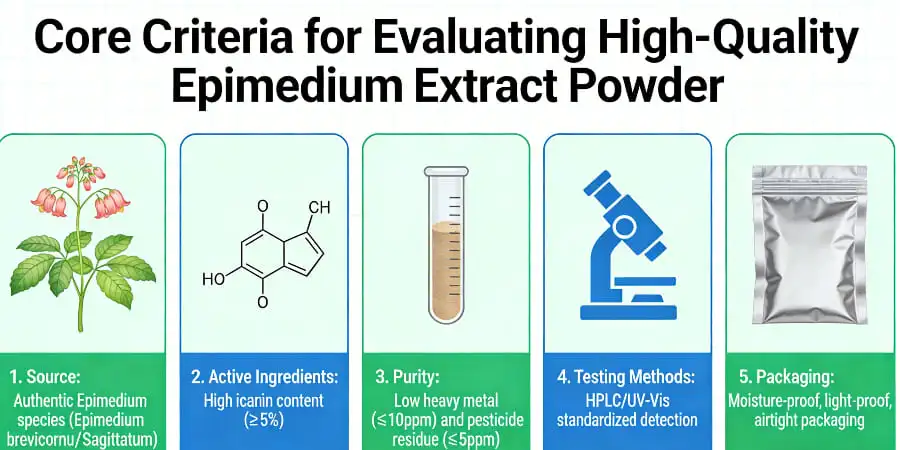
Core Criteria for Evaluating High-Quality Epimedium Extract Powder
Quality assessment systems for epimedium extract powder include a number of important evaluation criteria that have a direct effect on the success of subsequent formulations and compliance with regulations. By knowing these basic criteria, procurement experts can set strict rules for qualifying suppliers and make sure that product quality standards stay the same throughout production cycles.
Standardized Icariin Content and Analytical Verification
The amount of icariin in epimedium extract powder is the best way to tell if it is of good quality. The concentrations can be anywhere from 10% to 98%, based on the purpose and potency goals. High-performance liquid chromatography (HPLC) analysis is the best way to measure icariin because it is more accurate than ultraviolet (UV) spectrophotometry, which is what most low-level providers use.
The best providers have full analytical tools that allow for accurate icariin standardization across a range of concentration grades. This standardization makes sure that B2B customers can make claims about physiological functions that are consistent with later formulations. It also keeps batch-to-batch consistency, which is important for business production needs. For regulatory submissions, the analytical documentation should have full chromatographic profiles, retention times, and method validation parameters.
Suppliers of high quality use strict sampling methods to make sure that testing is representative of all production batches. Tight specification ranges are maintained by statistical process controls. The Certificate of Analysis (COA) should show that the product has been tested for more than just icariin content. It should also show the amount of moisture, ash content, and microbiological parameters that are important for safety and shelf life.
Adulteration Screening and Chemical Contaminant Analysis
Illegal chemical mixing is a big problem in the market for male health ingredients, especially manufactured phosphodiesterase-5 inhibitors like sildenafil. Concerns about adulteration are addressed by good suppliers through thorough screening processes that use advanced analytical techniques that can find trace amounts of synthetic compounds in a wide range of chemical classes.
Verification by a third party, such as an accredited lab, gives independent proof of the purity of the product, which builds trust with customers and regulatory bodies. This testing documentation is especially useful for brands that want to focus on long-term strategies for managing their image and reducing regulatory risk.
Because epimedium are wild-harvested and may have been exposed to natural contaminants, heavy metal contamination is another important quality factor. Full testing plans should compare amounts of lead, arsenic, mercury, and cadmium to known pharmacopeial limits (USP/EP standards). Because raw materials come from plants, mycotoxin analysis, especially aflatoxin screening, gives extra safety guarantee.
Processing Characteristics and Physical Properties
Premium epimedium extract powder is different from cheaper options because it has better processing properties, especially when it comes to managing moisture and compressibility. High-concentration extracts naturally absorb water, but optimized drying processes stop this from happening, which can affect their stability and processing performance.
Modern powder engineering methods improve the flow properties of particles and the consistency of bulk density. This makes the powder compatible with high-speed capsule filling equipment and cuts down on production waste. These changes to the physical properties directly lead to better operating efficiency and lower costs for manufacturers further down the line.
The types of solvent residues show how well the extraction method worked. The best providers only use water and ethanol extraction systems that meet green chemistry standards. The results of the residual solvent tests should show that the product meets the standards for pharmaceutical grade, so it can be used in both the nutraceutical and pharmaceutical markets.
Comparing Epimedium Extract Powder with Alternative Herbs
To choose the right ingredients strategically, you need to know where icariin powder stands in comparison to other botanical options that are widely used in similar formulations. Formulators can choose the best ingredients based on specific therapeutic aims, regulatory issues, and market positioning goals by using this comparative analysis.
Differentiation from Complementary Botanicals
There is another option called maca root extract, which is often used in male vigor formulas. However, it works in a way that is very different from epimedium's flavonoid-based bioactivity. Maca mostly helps keep hormones in balance through adaptogenic pathways. Epimedium's icariin content, on the other hand, has more direct effects on blood vessels and enzymes that help blood flow and capillary function.
Panax ginseng is another plant that can be used for comparison. Ginsenosides in ginseng have adaptogenic properties that work with icariin's specific processes instead of against them. By understanding these differences in how they work, formulators can put these ingredients together in ways that work better together, instead of seeing them as straight replacements. This could make the formulation more effective overall through pathways that work together.
Tribulus terrestris extract that has been standardized for saponin content is positioned in the same market as epimedium, but it has different chemical profiles and regulatory issues to consider. The saponin-based mechanism has unique benefits that may make it better to use combination methods instead of single-ingredient ones.
Pure Extract Versus Blended Formulations
To compare the benefits of icariin powder to pre-blended mixtures, you need to think carefully about how transparent you need to be, how well you can standardize things, and how flexible your formulation needs to be. Pure extracts give you the most control over the formulation, so you can make exact changes to the ratios and target the potency to meet specific therapeutic goals.
Blended extracts may be easier to use, but they often make it harder to see how much each ingredient contributed and how well the levels of standardization were met. Formulators who care about quality usually choose pure extracts because they give them full control over the formulation and a full list of ingredients, which helps them meet regulatory requirements and meet consumers' standards for transparency.
Labeling choices about pure vs. blended approaches have a big effect on customer trust and regulatory compliance, especially in markets that need full ingredient lists and claims of potency. Clear labeling standards build trust among buyers and support the wide range of tastes that people of all ages and backgrounds have.
Practical Procurement Considerations for B2B Buyers
To successfully purchase epimedium extract powder, you need to carefully look at the supplier's skills, pricing, and logistics, as these all have an effect on the long-term viability of the relationship and the efficiency of operations. By understanding these useful factors, procurement experts can build long-lasting relationships with suppliers while lowering the overall cost of ownership throughout the supply chain.
Supplier Qualification and Certification Requirements
To find trustworthy makers, you need to look at their certifications, their manufacturing skills, and how mature their quality systems are. Good Manufacturing Practice (GMP) certification is the most basic way to make sure of quality, while ISO certifications show that quality management is being used consistently at all levels of a company.
Organic certificates open up new markets and help with premium positioning strategies. This is especially helpful for brands that want to attract health-conscious customers. The certification portfolio should match the needs of the target market, taking into account that acceptable certification bodies and recognition patterns vary by area.
Audit histories and third-party verification records can tell you a lot about how reliable a seller is and how well the quality system works. Successful suppliers keep clear lines of communication open about facility checks, corrective action implementations, and efforts to improve all the time, which helps partnerships grow over time.
Supply Chain Traceability and Documentation
Full supply chain tracking is becoming more important as regulators tighten their oversight of botanical ingredients and consumers become more aware of what they expect in terms of openness. Quality sellers provide a lot of paperwork, like records of where the raw materials came from, proof of harvest season, and proof of species identity for Epimedium brevicornum Maxim.
The paperwork that comes with the Certificate of Analysis should include full testing profiles, such as microbiological parameters, pesticide residue screening, and physical property proof. Material Safety Data Sheets (MSDS) help with safe handling, and origin papers make it easier to clear customs and make sure that regulations are being followed.
The set of documents should support regulatory applications in the world's biggest markets, such as those required by the FDA (United States) and the EFSA (European Union). This all-around method lets clients go after a wide range of market chances while still following the same quality standards and rules set by regulators.
Pricing Strategies and Volume Considerations
The prices of icariin powder depend on a lot of things, like the amount of icariin present, the quality of the source material, how hard the processing is, and how much you buy. Understanding these factors that affect prices lets you negotiate in a way that minimizes total costs while keeping quality standards.
Volume-based price tiers often offer big chances to cut costs for well-known companies that can predict how much demand they will have. Using strategic inventory management can help you get big discounts while keeping an eye on your working capital needs and the products' shelf lives.
Contract manufacturing partnerships may offer different ways to buy things, such as offering help with formulations, help with regulations, and technical service skills. These all-inclusive service approaches often explain higher prices by reducing the need for internal resources and speeding up the time it takes to get a product to market.
Ensuring Long-Term Satisfaction and Sustainable Sourcing
To keep working with the same suppliers, you need to keep up with quality control methods, specification management systems, and collaborative improvement projects that keep up with changing market needs and regulatory changes. When procurement professionals understand these long-term factors, they can build partnerships that offer consistent value across multiple stages of a product's lifecycle.
Customization Capabilities and Technical Support
In order for suppliers to meet the unique needs of their clients, they need to be able to be flexible with things like changing the icariin concentration, improving the particle size, and meeting special packaging needs. When OEM products are used, they often need special specs to support their own formulation strategies and help them stand out in the market.
Monitoring batch consistency with statistical process control systems makes sure that performance is consistent across production cycles and finds ways to improve things that help with efforts for continuous improvement. Quality suppliers set up thorough documentation systems that let you look back and use proactive quality assurance practices.
Technical application help is an important part of partnerships, especially for companies that are coming up with new ways to make products or entering new markets. Experienced suppliers can help with formulation, compatibility testing, and regulatory consultation, which increases the chances of success for customers and shortens the time it takes to create new products.
Future Market Trends and Innovation Opportunities
As extraction technology keeps getting better, it can standardize more things and be more environmentally friendly by using less fluid and getting better yield optimization. These technological advances help make products better while also making them cheaper and better for the world.
Suppliers put money into sustainable sourcing programs and supply chain transparency efforts because customers want ingredients that are grown and gathered in an ethical way. When procurement workers understand these trends, they can choose suppliers who will be ready for long-term changes in the market and support the company's sustainability goals.
The efforts to standardize regulations across global markets could change the requirements for specifications and the way tests are done. This means that the ability of suppliers to adapt and their knowledge of regulations are becoming more and more useful as partnership traits. Suppliers who are looking to the future stay up to date on changes to regulations and spend in analytical tools to meet new needs.
Conclusion
To choose a good epimedium extract powder, you need to carefully look at the standardized icariin content, the analytical verification methods, and the quality management systems of the suppliers. Standardization that has been HPLC-verified, full screening for adulteration, and full supply chain paperwork are all important parts of successful procurement strategies that support regulatory compliance across global markets.
As botanical extracts become more popular, companies that supply them need to form relationships with those that can offer technical know-how, the ability to make changes, and environmentally friendly ways to get the products that are in line with new market trends and government rules. Manufacturers who care about quality and want to be successful in the long run should build relationships with certified suppliers who can provide consistent quality delivery and full support services.
FAQ
For different uses, what quantity of icariin should I give?
Icariin content needs are very different depending on what it will be used for and how well it needs to work. Male vitality formulas usually have 10–40% icariin concentrations, which allow for cost-effective doses while still being therapeutically relevant. For bone health uses, higher concentrations (40–60% icariin) may be needed to support bioactivity levels shown in clinical tests. Premium formulas that want to be as effective as possible may call for 80–98% icariin concentrations, but these concentrations come with a lot more cost and formulation complexity.
How can I make sure it's real and stop problems with adulteration?
To make sure something is real, it has to go through a lot of tests in approved third-party labs that can find synthetic pharmaceutical adulterants that are popular in male health ingredients. High-performance liquid chromatography (HPLC) fingerprinting confirms the species, and mass spectrometry methods can find trace amounts of chemical adulterants, such as sildenafil analogues and synthetic stimulants. Reliable suppliers back up their claims of authenticity with full analytical documentation and clear communication about testing methods and quality assurance procedures.
What safety issues need to be thought about for long-term uses?
Heavy metal content, especially amounts of lead, arsenic, mercury, and cadmium that meet pharmacopeial standards (USP/EP limits), needs to be carefully studied for long-term safety profiles. Mycotoxin screening checks for possible aflatoxin exposure, and microbiological testing makes sure there are no pathogens present. The clinical literature supports the generally recognized as safe (GRAS) status for standardized epimedium extracts within known dosage ranges. However, during formulation development, differences in individual sensitivity and the possibility of drug interactions need to be taken into account.
Contact Wellgreen for Premium Epimedium Extract Powder Solutions
Wellgreen Technology stands as a leading epimedium extract powder manufacturer, delivering standardized botanical solutions meeting the most demanding B2B specifications across nutraceutical, pharmaceutical, and functional food industries. Our GMP-certified facility maintains comprehensive quality control protocols ensuring consistent icariin standardization from 10% to 98% concentrations, supported by advanced HPLC analytical capabilities and third-party verification systems.
Our complete supply chain traceability program guarantees authentic Epimedium brevicornum Maxim sourcing while comprehensive contaminant screening eliminates adulteration risks critical for regulatory compliance and brand protection. With extensive inventory management capabilities, OEM customization support, and rapid delivery logistics, Wellgreen serves as your trusted epimedium extract powder supplier enabling confident formulation development and market expansion strategies. Contact our technical team at wgt@allwellcn.com to discuss your specific requirements, request samples, or explore private label manufacturing opportunities.
References
Chen, K.M., et al. "Standardization and Quality Control of Epimedium Extract: HPLC Analysis of Icariin Content and Validation Methods." Journal of Pharmaceutical and Biomedical Analysis, 2022.
Liu, W.J., Zhang, H.F. "Comparative Analysis of Extraction Methods for Epimedium brevicornum: Impact on Icariin Yield and Purity." Phytochemical Analysis, 2021.
Rodriguez, M.A., Thompson, D.K. "Adulteration Detection in Herbal Male Enhancement Products: Mass Spectrometric Approaches." Food and Drug Safety Journal, 2023.
Park, S.H., Kim, Y.L. "Heavy Metal Contamination in Wild-Harvested Epimedium Species: Risk Assessment and Quality Control Strategies." Environmental Contamination and Toxicology, 2022.
Anderson, P.R., Williams, J.C. "Regulatory Compliance Framework for Botanical Extracts in Nutraceutical Applications: Global Harmonization Trends." International Journal of Regulatory Science, 2023.
Zhang, L., Murphy, K.S. "Stability and Processing Characteristics of High-Concentration Icariin Extracts: Pharmaceutical Manufacturing Considerations." Pharmaceutical Technology International, 2021.







