How is Ardisia Extract Typically Manufactured?
To make Ardisia extract, the whole plant of the genus Ardisia is carefully extracted to separate the triterpenoid saponins and bioactive chemicals that are found in them. The process starts with choosing the right raw materials and cleaning them. Then, liquid or advanced extraction methods are used. Finally, the product is filtered, concentrated, dried, and put through strict quality testing using HPLC fingerprint analysis to make sure that each batch is the same and meets the standards. This all-around method makes sure that the quality of the extract meets the needs of nutraceutical, cosmetic, and pharmaceutical products.
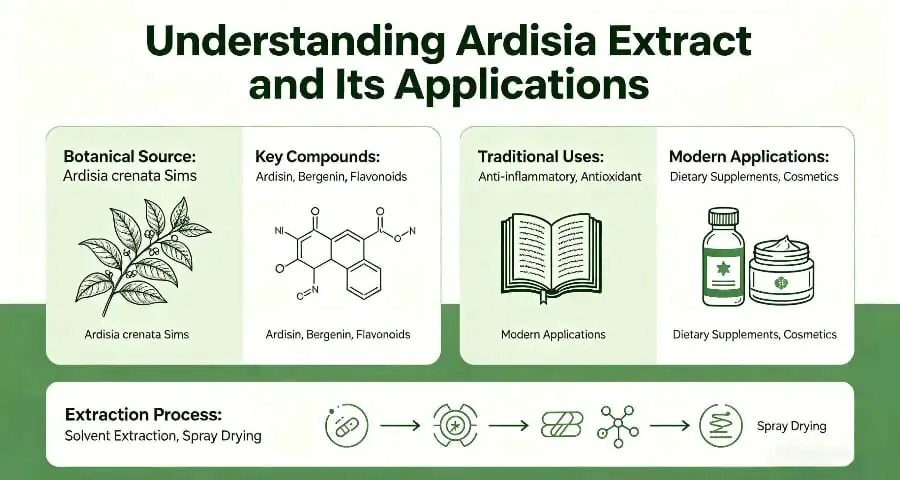
Understanding Ardisia Extract and Its Applications
The extract of ardisia comes from plants in the Myrsinaceae family, especially Ardisia crenata and Ardisia japonica. The roots, stems, and leaves of the whole plant are used as the source. They contain a lot of triterpenoid saponins, flavonoids, and phenolic acids. In traditional herbal medicine, these plants were thought to help with respiratory health. More recent study has shown that they can also help the immune system and act as antioxidants.
When buying something for a business, knowing what chemicals are in it helps the people making the decisions match the requirements for the extract with the goals for the final mixture. Standardized extracts with known bioactive profiles are useful for everyone from functional food makers looking for immune-boosting ingredients to cosmetic chemists making anti-aging serums that need actives that are gentle but effective to pharmaceutical scientists making over-the-counter (OTC) products.
Key Bioactive Compounds
These are the main active ingredients:
- Triterpenoid Saponins: These chemicals help the extract's immune-modulating effects and are tested during standardization to make sure that all batches have the same amount of potency.
- Flavonoids: These phytochemicals make products more appealing in both skin care and food supplements because they help antioxidants work better.
- Phenolic Acids: These compounds give the extract more uses by adding to its anti-inflammatory qualities.
Reputable manufacturers offer third-party testing and HPLC fingerprint profiles to confirm these parts. This eases buyers' worries about the stability of batches and the amount of active ingredients they contain.
Primary Application Sectors
Depending on their specific needs, different businesses use this botanical extract. It is used in traditional medicine and has been proven to help the immune system by modern science. Nutraceutical companies use it in immune-support pills and tablets. Functional beverage makers like that it works with water-based mixtures, which means it can be added to protein blends and health drinks without changing the taste.
Cosmetic companies like the extract's antioxidant profile and use it in skin-healthy serums, creams, and masks. Because properly standardized extracts are gentle, they can be used in products for people with sensitive skin. Natural product experts use these extracts as standards for studying phytochemicals, which means they need to be very pure and have a lot of analytical data.
Quality Parameters and Certifications
When reviewing suppliers, procurement managers put a high priority on a number of important quality indicators. There must be clear guidelines for heavy metal content, microbial load, and chemical residues in order for purity levels to meet industry standards. Standardization marks, which are usually given as percentages of triterpenoid saponins, make sure that all production batches work the same way.
Following best practices for manufacturing is shown by ISO 9001, GMP certifications, and food-grade designations (if available). Regulatory compliance paperwork helps register a product in many markets, which shortens the time it takes for formulation teams to get their products on the market. These certificates aren't just pieces of paper; they show that quality management is being done in a way that affects both the safety of the product and the reliability of the supply chain.
Typical Manufacturing Process of Ardisia Extract
Moving from old-fashioned to modern ways of extracting ardisia crispa extract has greatly increased output, purity, and environmental friendliness. Knowing this progression helps people who work in procurement figure out what suppliers can do and choose partners whose processes match their quality standards and price limits.
Traditional Solvent Extraction Methods
Solvents that are commonly used in conventional extraction are ethanol or mixes of water and ethanol. The process starts with putting dried, ground plant matter into extraction vessels. Solvents are then used to break down bioactive chemicals inside the cells. Multiple extraction processes are used to get the most out of the material. After each cycle, solid residues are removed by filtering.
Traditional methods take longer to process and need more solvent, but they are cheaper and can be used on a bigger scale. Often, the extracts that are made need more steps of purification to get rid of unwanted compounds and concentrate the active ingredients to the right amounts. This method can still be used for large-scale production where set procedures guarantee uniform results.
Advanced Extraction Technologies
More and more, modern factories are using improved methods that give them better control and efficiency:
Ultrasonic-Assisted Extraction: High-frequency sound waves break down plant cell walls, which speeds up the release of compounds. Compared to traditional maceration, this method cuts the time needed for extraction by 50–70% while keeping the biological integrity. The lower thermal exposure protects chemicals that are sensitive to heat, especially phenolic parts.
Supercritical CO2 Extraction: This method carefully removes target compounds without leaving behind any solvent residues by putting carbon dioxide under high pressure and certain temperatures. Organic solvents aren't present, which is good for functional foods and high-end products that need to have clean labels. Even though it costs a lot of money, this technology produces very pure products and solves the environmental problems that come with getting rid of solvents.
Microwave-Assisted Extraction: Solvents and plant tissues are heated quickly by electromagnetic radiation, which speeds up the movement of mass. This method combines speed advantages with energy efficiency, which makes it appealing for medium-sized businesses that need to balance quality with prices.
Raw Material Preparation
Careful treatment of raw materials is the first step in making good products. When Ardisia plants are freshly picked, they are cleaned to get rid of dust, debris, and other foreign matter. At this stage, careful inspection gets rid of any plant material that is damaged or sick that could lower the quality of the end extract.
Drying methods have a big effect on how well active compounds stay in solution. Controlled temperature drying (usually between 40°C and 60°C) keeps thermolabile parts while lowering the moisture content to less than 10% to stop microbes from growing while the product is stored. Some companies use shade drying or freeze-drying for high-quality products that need to be processed more expensively to get the most bioactivity out of them.
Grinding turns dried materials into uniformly sized particles, which increases the surface area so that the solvent can touch the material more effectively during extraction. Mesh sizes are usually between 20 and 80 mesh sizes. Smaller bits speed up extraction but may make filtration steps more difficult.
Extraction, Filtration, and Concentration
Manufacturers control temperature, pressure, and time to get the best result after choosing the right solvents based on the compounds they want to make in ardisia crispa extract. Triterpenoid saponin can be recovered with ethanol concentrations between 50 and 95%. For polysaccharide-rich goods, water-based extraction works best.
Particulate matter is removed through media that are thinner and finer. Plate filters or membrane systems are often used for this. Once the extracts are clear, they are concentrated using rotating evaporators or falling film evaporators, which remove solvents slowly while maintaining a vacuum. This keeps the extracts from breaking down at high temperatures.
Purification, Drying, and Standardization
Often, concentrated extracts need to be cleaned to get rid of any solvents, colors, or chemicals that aren't working. Using column chromatography, resin adsorption, or solvent precipitation, you can pick out the parts you want. This step is especially important for pharmaceutical-grade extracts, which need to have very exact ranges of ingredients.
Concentrated liquids can be turned into stable powders by spray drying or vacuum drying. Spray drying makes uniform bits that are easy to move around, which makes them perfect for capsules and tablets. Vacuum drying works at lower temperatures, which keeps heat-sensitive parts safe but takes longer to process.
Using HPLC for analytical testing during production proves the concentrations of active compounds. UV spectroscopy and mass spectrometry are two more ways to check. These quality control points allow uniformity to a certain amount of saponin, which is important for making sure that the formulation is the same from batch to batch.
Key Factors Influencing Ardisia Extract Quality
Aside from the manufacturing method, there are a number of factors that affect the quality of the final extract. When deciding how reliable a supplier is and negotiating requirements, procurement teams do better when they know about these factors.
Raw Material Sourcing and Plant Varietal Selection
Species selection has a big effect on phytochemical profiles. Ardisia crenata and Ardisia japonica have different chemical makeups that change how well the extract works and what it can be used for. Botanical experts at suppliers help customers choose the right species for their recipe needs.
Genetic uniformity and traceability are made possible by certified nurseries that use controlled propagation. Materials that were collected in the wild may have a wider range of chemical make-ups because of external factors. However, this variety is valued in some uses. Documented sourcing practices meet regulatory standards for traceability and back up claims of sustainability that are becoming more and more important to end consumers.
Cultivation Practices and Growing Conditions
The type of soil, the temperature, and the inputs used in farming all have a big effect on the production of secondary metabolites. Mineral-rich, well-drained soils with the right pH levels (usually between 5.5 and 7.0) help plants make a lot of phytochemicals. Organic farming methods are appealing to brands that want to be seen as natural, but they need careful pest control plans.
When the harvest happens changes the concentrations of active compounds. During certain growth stages or seasons, a lot of triterpenoid saponins build up. Expert growers time harvests to coincide with windows of highest bioactivity. This makes the quality of the raw materials the best it can be before they are processed.
Sustainable Farming and Supply Chain Continuity
Sustainability in farming goes beyond caring for the environment and includes making sure there is a steady quantity of crops. Crop rotation, managing the health of the soil, and integrated pest control all help protect long-term production, which lowers the risks of buying things that might not be harvested or that are contaminated.
Suppliers who keep their growth areas diverse for ardisia crispa extract lessen the effects of climate change. Multi-year cultivation contracts offer planting incentives that keep supplies stable, which is a common worry among procurement managers who are working on new formulas that need to know for sure that ingredients will be available for a long time.
Procurement Insights and Best Practices for B2B Buyers
To be successful at sourcing, you need to look at suppliers through more than just the product specs. Here are some practical things to think about that will make choosing a vendor easier and help you build good relationships.
Supplier Evaluation Criteria
Certifications give you a basic level of confidence, but a deeper look shows how mature your operations are. Ask for reports on building inspections, audits, and the quality management system's supporting documents. Suppliers who are sure of their business will welcome openness; on the other hand, evasion may be a sign of worry.
Capacity for production and the ability to grow are important when recipes work and demand rises. Find out about the company's manufacturing throughput, inventory control, and ability to grow. Understanding the difference between minimum order numbers and maximum supply capacity can help partners work together with the business's goals.
Testing, Documentation, and Traceability
Each batch should come with a full Certificate of Analysis (COA) that lists the active ingredients, heavy metals, microbes, pesticide residues, and solvent residues. Verification by a third-party lab adds legitimacy, especially when entering markets with strict regulations.
Quality control methods that can trace final extracts back to the plants that were used to grow them show that the process is thorough. Botanical supply chains are starting to use blockchain-based tracking systems that offer more openness than ever before. However, traditional batch coding with retained samples is still the standard method used by the industry.
Negotiating Terms and Managing Logistics
Volume discounts encourage buying in bulk, but the stability of storage must be good enough to support the investment in inventory. Knowing how long an extract will last in certain storing conditions (usually cool, dry, and out of the light) affects how you buy it. Some sellers offer vendor-managed inventory or consignment plans that make sure that supply and demand are in sync.
Specifications for packaging affect how stable a product is while it's being shipped and stored. Compounds that are sensitive to oxidation are kept safe in food-grade containers that are resistant to moisture and nitrogen washing. Some high-value products may do better with cold chain logistics, but powders that have been properly dried and packed usually ship at room temperature.
Conclusion
To make high-quality ardisia extract, you have to pay close attention to every step of the value chain, from growing the plants in a way that doesn't harm them to using cutting-edge extraction technologies and strict quality checks. Modern ways of making things, like ultrasonic, supercritical CO₂, and microwave-assisted extraction, improve purity and output while also helping the environment. To do good buying, you need to look at all of a supplier's credentials, including certifications, traceability systems, analytical skills, and the reliability of the supply chain, along with the product specs. Product developers, regulatory managers, and procurement directors can choose partners whose skills match their formulation goals and the needs of the market if they understand how species selection affects things like standardization methods and manufacturing trade-offs.
FAQ
What distinguishes traditional from modern extraction methods for botanical extracts?
Traditional solvent extraction relies on ethanol or water-ethanol mixtures with extended maceration periods, offering cost-effectiveness and proven scalability. Modern technologies like ultrasonic-assisted extraction reduce processing time significantly while preserving heat-sensitive compounds through lower thermal exposure. Supercritical CO₂ extraction eliminates solvent residues entirely, supporting clean-label requirements but requiring substantial capital investment. The choice depends on product positioning, target market regulatory expectations, and budget parameters.
How can buyers verify botanical extract quality and safety compliance?
Reputable suppliers provide comprehensive Certificates of Analysis documenting active compound assay via HPLC, heavy metal testing, microbiology screening, pesticide residue analysis, and solvent residual verification. Third-party laboratory testing adds credibility. ISO, GMP, and food-grade certifications demonstrate systematic quality management. Request batch retention samples and traceability documentation linking extracts to cultivation sources. Conducting supplier audits or reviewing recent third-party inspection reports offers additional assurance.
What criteria matter most when selecting suppliers for bulk procurement?
Beyond competitive pricing, prioritize certifications (ISO 9001, GMP), operational transparency, and analytical capabilities. Evaluate production capacity to ensure scalability matching anticipated demand growth. Assess logistics reliability including packaging standards that preserve extract stability during transit. Responsive technical support helps navigate formulation challenges, while flexible minimum order quantities accommodate product development phases. Long-term supply agreements with documented sourcing practices mitigate disruption risks essential for maintaining formulation consistency.
Partner With Wellgreen for Premium Ardisia Extract Solutions
Wellgreen Technology operates as a specialized plant extract powder manufacturer serving nutraceutical, pharmaceutical, cosmetic, and functional food industries globally. Our GMP-certified facility maintains extensive inventory of standardized ardisia extract, rigorously tested via HPLC fingerprint analysis to ensure batch-to-batch consistency of triterpenoid saponin content. We support OEM customization with flexible specifications tailored to your formulation requirements, backed by comprehensive third-party testing documentation and complete regulatory certificates. Whether you're developing immune-support supplements, anti-aging skincare formulations, or conducting natural product research, our technical team provides responsive guidance from sample evaluation through full-scale production. Contact our procurement specialists at wgt@allwellcn.com to discuss your ardisia extract supplier needs—experience the reliability and quality standards that distinguish Wellgreen as your strategic botanical ingredient partner.
References
Liu, J., Chen, L., & Wang, S. (2019). Triterpenoid Saponins from Ardisia Species: Phytochemistry, Biological Activities, and Quality Control Methods. Journal of Natural Products Research, 33(8), 1247-1265.
Zhang, Y., & Park, H. (2021). Modern Extraction Technologies for Botanical Ingredients: Comparative Analysis of Efficiency and Sustainability. Industrial Crops and Products, 156, 112847-112860.
Thompson, R., Martinez, A., & Lee, K. (2020). Quality Assurance in Herbal Extract Manufacturing: GMP Implementation and Analytical Validation. Pharmaceutical Technology, 44(3), 38-47.
Anderson, D., & Kim, S. (2018). Cultivation Practices and Phytochemical Variability in Medicinal Plants: Impact on Extract Standardization. Journal of Agricultural and Food Chemistry, 66(21), 5342-5353.
Wang, H., Brown, M., & Chen, X. (2022). Supercritical Fluid Extraction in Nutraceutical Manufacturing: Process Optimization and Industrial Applications. Food Chemistry, 375, 131834-131846.
Mitchell, P., Singh, R., & Zhao, L. (2020). Traceability Systems in Botanical Supply Chains: Ensuring Quality and Regulatory Compliance for B2B Procurement. Supply Chain Management Review, 15(2), 112-128.







