How to Use Horse Chestnut Extract?
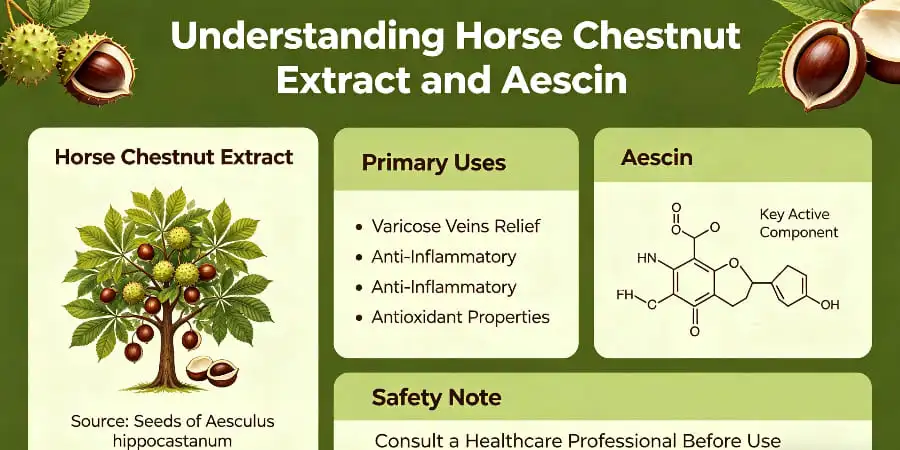
To get the most out of horse chestnut extract, you need to know about its main beneficial ingredient, aescin escin (also written as escin), a triterpenoid saponin complex that is taken from the seeds of Aesculus hippocastanum. This white to off-white powder works by making capillary walls stronger, lowering the permeability of blood vessels, and stopping the activity of the hyaluronidase enzyme. In real life, standardized extracts with 16–20% aescin are made into supplements that people take by mouth (usually 300–600 mg per day), creams that are put on the skin to keep veins healthy, and cosmetics that target swelling and circulation. Purchasing managers should give more weight to suppliers who offer consistent batch-to-batch total saponin content, proof of third-party testing, and specifications that are specific to the dosage forms that will be used, such as pharmaceutical-grade APIs, food-grade supplement powders, or cosmetic-grade actives.
Understanding Horse Chestnut Extract and Aescin (Escin)
The Chemistry Behind the Active Compound
There are at least 30 different types of triterpenoid saponins that make up aescin. The two main types are beta-aescin (which is physically active) and alpha-aescin. Its complex glycosidic structure is shown by the molecular formula C55H86O24. This structure helps it dissolve in both water and alcohol, which is a useful property for a variety of preparation needs. Unlike other plant saponins, this particular mix allows for targeted pharmacological action at the level of the vein wall.
To get pure extracts from horse chestnut seeds that can be used in medicinal and food-grade products, it is important to carefully remove any harmful substances, especially esculin. Standardized standards from good suppliers range from 20% to 98% total saponin content, and pharmaceutical-grade materials usually keep purity levels at least 95%. This precision is necessary so that formulators can figure out exact doses and make sure that clinical results are the same from one production batch to the next.
Mechanism of Action and Therapeutic Benefits
Aescin's venotonic qualities come from its ability to seal capillary membranes by stopping the breakdown of proteoglycan in vessel walls. This process directly deals with vascular permeability, which is what causes tissues to hold water. Aescin stops the leakage of low-molecular-weight proteins, electrolytes, and water into interstitial spaces. This successfully gets rid of edema and the feeling of having heavy legs that comes with chronic venous insufficiency.
The substance also reduces inflammation in a number of ways, such as by blocking prostaglandins and acting as an antioxidant. Because of these qualities, it is very useful for making medicines that treat post-operative swelling and hemorrhoids. Studies that have been published in peer-reviewed journals show that people who take standard aescin doses of 100 to 150 mg every day see changes in their venous tone and microcirculation parameters.
Applications Across Industrial Sectors
High-purity aescin is used as an active ingredient (API) in tablets, gels, and injections that are used to treat vascular diseases. Because the compound has been shown to strengthen capillaries, it can be used in formulas to treat varicose veins, phlebitis, and hematomas after injuries. Regulatory approval in European markets sets an example for regulations for quality standards and clinical validation.
Nutraceutical companies put standardized horse chestnut extract in products that are sold to help the circulatory system. Each serving of these products usually has 300 to 600 mg of extract, which is standardized to give 50 to 120 mg of horse chestnut extract. Because the compound is safe at approved doses, it gives formulators a lot of freedom when making products that work on their own or with other ingredients that support blood vessels, like diosmin or hesperidin.
Aescin's anti-edema features are used in cosmetics to make eye care products that reduce dark circles and puffiness. The ingredient is also used by beauty brands in slimming products and cellulite treatments because it helps the lymphatic system and lowers the amount of fluid that stays in one area. Cosmetic chemists usually work with specs of 5 to 20 percent total saponins, which are tailored to work well on the skin and in topical delivery methods.
How to Use Horse Chestnut Extract Effectively?
Dosage Guidelines Based on Clinical Evidence
To get the desired results while staying safe, therapeutic applications need to follow dosing guidelines that are based on evidence. Most clinical studies that back up claims about vascular health use 300 to 600 mg of standardized extract every day, which is equal to 100 to 150 mg of aescin. This range has shown big improvements in reducing leg swelling, relieving pain, and managing edema in people with chronic venous insufficiency.
Topical products have smaller amounts of extract, usually between 1% and 5% in creams, gels, and serums. These concentrations provide anti-inflammatory benefits in specific areas without causing worries about systemic exposure. Highly purified aescin sodium salt is used in controlled hospital settings to make pharmaceutical injection preparations. However, supplement or cosmetic makers don't usually buy this kind of specialized product.
Bioavailability can change between different types of extract, so people who make products need to take this into account. Enteric coating or liposomal delivery methods help oral preparations get absorbed better and keep the saponin complex from breaking down in the stomach. When working with aescin powder in topical matrices, cosmetic formulators should think about using penetration enhancers or nano-emulsion technologies to get the most out of skin delivery.
Comparing Dosage Forms and Their Industrial Applications
Standardized powder extracts give B2B buyers in a wide range of industries the most options. These powders don't stick to anything and work well with processes like filling capsules, pressing tablets, and adding vitamins to drinks. Because aescin is water-soluble, it is easier to spread in water-based systems. However, formulators should be aware that saponin compounds can foam when they are mixed at high pressure.
It's easier to use liquid extracts, like hydroalcoholic tinctures or glycerin-based solutions, for certain tasks, like making tinctures or ready-to-drink useful drinks. These mixes don't need any dissolving steps, but the amount of alcohol they contain needs to be carefully thought out, especially in markets that like clean labels or where the government limits the amount of ethanol that can be in dietary supplements.
Microencapsulated or spray-dried forms can help with stability issues in situations where wetness is a problem. These special forms keep aescin from breaking down due to oxygen and make it easier for direct-compression tablet formulations to flow. These value-added forms are often preferred by contract makers who work with the functional food industry to make sure consistent dispersion in protein powders, energy bars, and fortified snacks.
Formulation Compatibility and Stability Considerations
Since aescin is very stable in neutral to slightly acidic pH ranges (4.5–7.0), it can be used in most supplement and makeup formulations. Formulators should stay away from conditions that are very alkaline, as these can break down saponin and make the product less effective over time. When making something, temperature sensitivity needs to be thought about; long-term contact above 60°C can weaken bioactivity, especially in systems that don't contain water.
Bioflavonoids like diosmin, hesperidin, and rutin have been shown to work in synergy with horse chestnut extract when combined with other ingredients that strengthen the blood vessels. These mixes make the venotonic effect stronger overall while letting smaller doses of each ingredient be used. Antioxidants like vitamin C or E help protect the saponin complex from oxidative stress while it is being stored. This makes the product more stable when used topically or orally.
When cosmetic scientists work with aescin powder, they need to make sure it works well with the emulsifiers, preservatives, and active ingredients that are used in their particular formulations. Saponins' surfactant qualities can change the stability and texture of an emulsion, so thickening agents and co-emulsifiers need to be optimized. Patch testing is still needed to make sure that the skin can handle the product, especially for leave-on goods that are made with higher concentrations.
Procuring Horse Chestnut Extract for Your Business
Evaluating Supplier Credentials and Certifications
A successful procurement process starts with a careful check of the supplier's quality control systems and compliance with regulations. ISO 9001 certification shows that quality processes have been formed, and ISO 22000 or FSSC 22000 certification shows that food safety management skills are applicable to supplement-grade materials. For pharmaceutical applications, GMP certification must be in line with regional standards. For example, FDA registration is needed for U.S. markets, EU GMP certification is needed for European distribution, or similar standards must be met in target jurisdictions.
Ask for thorough descriptions of the manufacturing process to learn about solvent systems, extraction methods, and purification methods. The standard in the business is ethanol-water extraction, but some suppliers use their own special methods that they say make the product more bioavailable or lower in impurities. Trace solvent residue testing should be done to make sure that the limits set by the pharmacopeia are met, especially if the goods being made are subject to strict regulatory oversight.
Auditing skills depend on how big and complex the supplier is. Large manufacturers usually allow on-site facility reviews, which give people a clear picture of the work environments, how equipment is calibrated, and how employees are trained. As an alternative, smaller suppliers might give virtual tours or audit reports from a third party. A look at the documentation, which includes batch records, cleaning validation, and change control processes, shows how mature and consistent the operations are.
Understanding Grade Distinctions and Specifications
Material types have a big effect on both how well they work in a given application and how much they cost. Pharmaceutical-grade aescin powder that is standardized to 95% total triterpene saponins is used in high-potency supplement formulations and API needs. A lot of tests are done on these products, such as heavy metal analysis (for example, lead, arsenic, cadmium, and mercury) and microbiological screening to Ph.Eur. or USP standards.
Nutraceutical-grade extracts usually have between 20 and 50 percent total saponins. Lower concentrations are enough for normal supplement uses and are cheaper. These materials keep up quality standards that meet GMP requirements for dietary supplements while lowering the costs of raw materials in areas where prices are important. Formulators should make sure that the methods used for standardization are in line with the calculations used for label claims and the testing processes for finished products.
Cosmetic-grade standards stress both chemical cleanliness and compatibility with the skin. For leave-on topical treatments, preservative systems, residual solvents, and microbial limits get more attention. Some suppliers offer water-soluble aescin derivatives or pre-dispersed formats that are especially designed for cosmetic uses. This makes it easier to make formulations that include powder extracts in emulsion systems.
Negotiating Volume Pricing and Supply Agreements
Prices on the market depend on many things, such as the supply of raw materials, the rate of extraction, the purity requirements, and the number of orders. Pharmaceutical-grade materials are more expensive because they need to be tested more thoroughly and are only made in small quantities. Supplement-grade products are more cost-effective, and when you order 100 kg or more, you can usually get a big discount.
People who buy and people who sell things regularly both benefit from long-term supply deals. Committed volume forecasts help suppliers plan production more efficiently and keep enough raw materials on hand, which leads to better prices and guaranteed supply. Buyers get price insurance against changes in the market and get first choice when supply is limited. These kinds of partnerships work best when both sides are open and honest about their predictions of demand and their output capabilities.
Different providers have very different payment terms, lead times, and minimum order amounts. Smaller trial orders (5–25 kg) are often accepted by established manufacturers with a lot of inventory space to help with product development. However, the per-unit price represents the problems that come with ordering in bulk. Direct ties with manufacturers get rid of markups for distributors, but they also require bigger minimum orders. Distributor partnerships give companies that are still in the early stages of growth more options and faster delivery.
Conclusion
Horse chestnut extract, which has been tested for its aescin escin content, is a scientifically proven ingredient that can be used in many different ways in the cosmetics, nutraceuticals, and pharmaceutical industries. To use the compound effectively, you need to know how it works, choose the right specs for the job, and work with qualified suppliers who can provide consistent quality and technical support.
A successful procurement depends on carefully evaluating suppliers, being clear about what is needed, and building smart relationships with them to ensure a steady supply and the lowest possible costs. Horse chestnut extract is a good addition to product lines that aim to support the circulatory system, reduce inflammation, and treat edema on the skin because of its effectiveness in addressing vascular health issues, as well as its favorable safety profile and clean label positioning.
FAQ
What is the difference between aescin and escin?
The terms aescin and escin refer to the same bioactive saponin complex extracted from horse chestnut seeds. This naming variation stems from transliteration differences across languages and scientific literature. Both designations describe the mixture of triterpenoid glycosides responsible for the extract's venotonic and anti-inflammatory properties. Procurement specifications may use either term interchangeably, though pharmaceutical compendia and European suppliers more commonly employ "aescin" while some U.S. sources prefer "escin."
How do I select the right supplier for pharmaceutical-grade horse chestnut extract?
Prioritize suppliers holding GMP certifications aligned with your target markets and offering comprehensive analytical testing including HPLC saponin profiling, heavy metal screening, microbial limits testing, and residual solvent analysis. Review Certificates of Analysis from multiple production batches to assess consistency. Request stability data supporting your product's intended shelf life. Evaluate technical support capabilities and willingness to provide regulatory documentation. Consider conducting supplier audits or reviewing third-party audit reports to verify manufacturing controls and quality systems.
What specifications should I request for nutraceutical applications?
Request standardized extracts containing 16-20% total triterpene saponins for typical supplement formulations, ensuring specifications include identity confirmation via HPLC, heavy metal limits (typically <10ppm total, <3ppm lead), microbial counts meeting supplement GMP standards, and pesticide screening. Verify the supplier provides batch-specific Certificates of Analysis, allergen declarations, and GMO status documentation. Confirm particle size distribution if directly encapsulating powder, and request flow properties data if using automated filling equipment.
Partner with Wellgreen for Premium Horse Chestnut Extract Supply
Wellgreen Technology stands as your trusted horse chestnut extract supplier, delivering pharmaceutical-grade and nutraceutical-grade aescin escin powder backed by comprehensive quality certifications. Our GMP-certified manufacturing facility maintains extensive inventory of standardized extracts (20%-98% total saponins), ensuring rapid fulfillment for both development samples and commercial-scale orders.
We provide complete testing documentation including third-party verification, stability data, and regulatory support materials to streamline your product development and compliance processes. Our technical team offers formulation guidance tailored to your specific application—whether developing oral supplements, topical treatments, or cosmetic formulations. With flexible MOQ options, OEM customization capabilities, and strict quality control at every production stage, Wellgreen delivers the reliability your supply chain demands. Contact our procurement specialists at wgt@allwellcn.com to request samples, specifications, and customized quotations for your next project.
References
Sirtori CR. Aescin: pharmacology, pharmacokinetics and therapeutic profile. Pharmacological Research. 2001;44(3):183-193.
Pittler MH, Ernst E. Horse chestnut seed extract for chronic venous insufficiency. Cochrane Database of Systematic Reviews. 2012;11:CD003230.
Dudek-Makuch M, Studzińska-Sroka E. Horse chestnut – efficacy and safety in chronic venous insufficiency: an overview. Revista Brasileira de Farmacognosia. 2015;25(5):533-541.
Guillaume M, Padioleau F. Veinotonic effect, vascular protection, antiinflammatory and free radical scavenging properties of horse chestnut extract. Arzneimittelforschung. 1994;44(1):25-35.
Bombardelli E, Morazzoni P, Griffini A. Aesculus hippocastanum L. Fitoterapia. 1996;67(6):483-511.
European Medicines Agency. Assessment report on Aesculus hippocastanum L., semen. Committee on Herbal Medicinal Products. 2009;EMA/HMPC/638244/2008.







